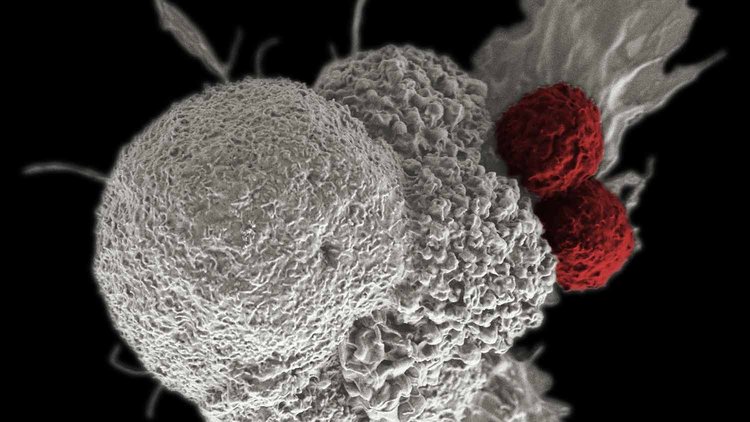
For years, immunotherapy—a treatment intended to bolster the immune system to eradicate cancer—was an exciting idea that had not lived up to its promise.
Until recently, immunotherapy was “not very successful at all,” said James P. Allison, PhD, chair of the Department of Immunology at The University of Texas MD Anderson Cancer Center in Houston. “That was one of the reasons it was kind of held in disrepute.”
But attitudes changed in the last decade because of the success of clinical trials of monoclonal antibodies, such as ipilimumab (trade name Yervoy), which was approved by the Food and Drug Administration (FDA) in 2011 for the treatment of melanoma. The development of ipilimumab was based on Allison’s work while he was director of the Cancer Research Laboratory at the University of California, Berkeley.
“We’re past the stage where we say, ‘Oh, this next thing will take it over the top,’ because ‘over the top’ is where we are now. There are tens of thousands of people alive who wouldn’t be otherwise.”
James P. Allison, PhD
University of Texas MD Anderson Cancer Center
Fast forward to August 2017. The FDA approved a cancer therapy, developed by researchers at the University of Pennsylvania, that genetically engineers a patient’s T cells to more effectively attack leukemia cells and, in some patients, eradicate the disease. The technology, chimeric antigen receptor (CAR) T, is approved specifically for the treatment of refractory B-cell acute lymphocytic leukemia. This therapy is also showing promise in treating chronic lymphocytic leukemia and non-Hodgkin lymphoma.
“We’re past the stage where we say, ‘Oh, this next thing will take it over the top,’ because ‘over the top’ is where we are now,” said Allison, also a primary inventor of a kind of immunotherapy known as checkpoint blockade. “There are tens of thousands of people alive who wouldn’t be otherwise.”
Other researchers agree that immunotherapy has turned the corner.
“I think it’s been dramatically successful. Patients are achieving complete regression of their cancers. And there’s a significant percentage of these patients who have very long remissions and are probably cured,” says Gerald P. Linette, MD, PhD, chief medical officer for Cancer Immunotherapy at Penn Medicine and professor of medicine at the Hospital of the University of Pennsylvania in Philadelphia.
“Historically our role [in academic medicine] has been more in early discovery or basic research—getting funding from the NIH to study a very fundamental aspect related to the immune system,” said Linette. “But I would say at Penn and a few other places around the country that have the infrastructure, we can actually take it one step further and do real translational research … we take a good idea from the lab and because of the facilities and expertise can actually develop our own treatments, independent of industry.”
What is immunotherapy?
Many of us live with mutations that lead to cancer. But tumors never develop because our ever-watchful immune systems identify the mutations, mobilize antibodies and leucocytes, and clear the malignant cells.
Sometimes, however, our immune systems are overwhelmed or fail to respond. Immunotherapy is designed to help the immune system by revving up its response or making the cancer cells more vulnerable.
A time-tested precursor to modern immunotherapy is bone marrow transplantation, developed first in the 1950s at the Fred Hutchinson Cancer Research Center in Seattle, Wash. Cancer vaccines such as BCG (bacille Calmette-Guerin) and interferon alfa and interleukin-2 have been used to stimulate immune systems. But today, two newer forms of immunotherapy are generating the most excitement: checkpoint inhibitors and CAR T- cells.
Checkpoint inhibitors, said Allison, “take the brakes off the T cells,” the specialized class of white blood cell that intercept and destroy intruders such as viruses and mutant cells. Cancer cells often defend themselves by displaying surface molecules that latch onto a molecular receptor on the T cells. Blocking this “checkpoint” turns the T cells off and allows tumor cells to avoid immune destruction.
Researchers are finding checkpoint inhibitors to block these interactions and allow the T cells to continue their attack. The FDA approved this therapy, referred to as “checkpoint blockade” for melanoma in 2011. This is the treatment former president Jimmy Carter received for melanoma that had metastasized. After having surgery and radiation therapy as well, Carter announced in 2015 that his cancer was undetectable.
“There’s still a lot of basic research and laboratory research that needs to take place to fulfill the promise and potential of cancer immunotherapy. We need more [financial] investment combined with higher rates of clinical trial participation.”
Gerald P. Linette, MD, PhD
Hospital of the University of Pennsylvania
Alison conducted a randomized placebo-controlled trial with patients in late-stage melanoma. “Of the patients who were treated, about 20% of them are alive four years after they were treated,” reported Allison, a pioneer in developing checkpoint inhibitors. Two prominent checkpoints on T cells are PD-1 and CTLA-4. “If you give patients the two drugs, one targeting CTLA-4 and the other targeting PD-1, about 60% [of melanoma patients] survive at least two years later,” he continued. “They’re basically cured.”
New approvals of checkpoint blockades followed for treatment of lung cancer, bladder cancer, some types of colon cancer, kidney cancer, and Hodgkin lymphoma, said Allison. In May 2017, the FDA approved a PD-1 blockade (with the monoclonal antibody pembrolizumab, trade name Keytruda) for several cancers—including colorectal, endometrial, and gastrointestinal—with a common biomarker. This is the first time an immunotherapy treatment was based on a common biomarker for different kinds of cancer.
With CAR T, another remarkable immunotherapy, T cells are drawn from the patient and engineered to produce chimeric antigen receptors on their surface. The CAR proteins allow T cells to recognize an antigen on tumor cells. The engineered cells are multiplied to the millions and infused back into the patient. The cells attack the cancer and remain in the body long after treatment to continue their attack on malignant cells.
Immunotherapy may have advantages other therapies don’t.
“[With immunotherapy] you’re not attacking the tumor directly, so unlike chemotherapy or the small molecules [that penetrate the cancer cell membrane], you don’t really need to know what is causing the tumor,” said Allison. And once you’ve got the cells—genetically modified T-cells or checkpoint blockade—“you’ve got memory,” he continued. “Once you get an immune response started, the immune system itself can take care of new antigens that appear [for the rest of a person’s life].”
Thanks to academic medicine
Academic institutions are in a good position to study the whole panoply of problems and come up with novel solutions, noted Ross McKinney Jr., MD, AAMC chief scientific officer. “These are difficult, complicated problems that require a variety of different strategies.”
“Some of our institutions have been the real pioneers in the immunotherapy world,” McKinney observed. “That’s the role of the academic medical center—to generate the idea, to demonstrate that it has potential, and then, because we are not in the business of selling drugs and therapies, we license or hand it off to corporate partners that are used to doing that kind of work.”
Despite rapid recent advances in immunotherapy, Linette noted, “We’re nowhere near close to successfully treating all patients that walk through the door. There’s still a lot of basic research and laboratory research that needs to take place to fulfill the promise and potential of cancer immunotherapy [for people with all cancers]. We need more [financial] investment combined with higher rates of clinical trial participation.”